You are working a busy emergency department shift when a 52-year-old woman rolls in tachycardic to 148, febrile, diaphoretic, and agitated. Her ECG shows rapid atrial fibrillation. The team is already reaching for adenosine when you notice her hands are trembling and her neck looks full. The reflex is there — treat the rhythm. But experienced emergency clinicians know that in this scenario, treating the cardiac presentation without identifying the underlying endocrine driver can be catastrophic.
Thyroid storm and adrenal crisis are two endocrine emergencies that routinely present with cardiac manifestations so convincing that they are frequently misattributed to primary cardiac disease. Both carry significant mortality when missed — thyroid storm kills 5 to 25 percent of patients without aggressive intervention, and adrenal crisis-related hemodynamic collapse can be indistinguishable from cardiogenic shock at first glance. Understanding how to differentiate these conditions from true cardiac emergencies is not just academic; it is a core clinical survival skill.

In this article, we walk through the pathophysiology, clinical presentations, diagnostic clues, and emergency management of both thyroid storm and adrenal crisis, with a specific focus on how these conditions mimic cardiac events and what features help you distinguish them. We also explore how these emergencies connect directly to the ACLS framework you rely on every shift.
Thyroid storm — also called thyrotoxic crisis — represents the extreme end of uncontrolled hyperthyroidism. It is a clinical diagnosis defined by severe, life-threatening exaggeration of thyroid hormone effects on multiple organ systems. The cardiovascular system bears the brunt of this hormonal assault, which is exactly why thyroid storm so convincingly mimics primary cardiac emergencies.
Thyroid hormones (T3 and T4) exert direct and indirect effects on cardiac function. T3 increases expression of myosin heavy chain isoforms, enhances cardiac contractility, and reduces systemic vascular resistance — collectively producing a hyperdynamic circulatory state. In thyroid storm, this effect is amplified dramatically. Heart rate rises due to both direct chronotropic stimulation and heightened sensitivity to catecholamines. Cardiac output can double or triple from baseline. The combination of tachycardia, high output state, and increased myocardial oxygen demand sets the stage for dysrhythmia, heart failure, and in severe cases, cardiovascular collapse.
Atrial fibrillation is present in up to 35 percent of thyroid storm cases, making it the most common arrhythmia encountered. This is not coincidental — thyroid hormones shorten the atrial refractory period and increase automaticity, creating the perfect substrate for atrial fibrillation. When an emergency provider sees rapid AFib and does not identify a precipitating cause, thyroid storm must be high on the differential. For a deeper understanding of atrial fibrillation mechanisms and management, see our guide on Understanding Atrial Fibrillation: Causes, Symptoms, and Treatments.
The cardiovascular manifestations of thyroid storm include tachycardia exceeding 140 beats per minute, wide pulse pressure, hyperdynamic precordium, and signs of high-output heart failure including pulmonary edema and peripheral edema. When a patient presents with rapid atrial fibrillation plus pulmonary edema, the knee-jerk assumption is acute decompensated heart failure with an underlying cardiac etiology. Add in ST-segment changes from demand ischemia and now the differential list fills up with ACS, STEMI equivalents, and cardiomyopathy — all while the real driver is a thyroid gland in full crisis.
Wide complex tachycardia can also occur in thyroid storm due to rate-related aberrancy or underlying Wolff-Parkinson-White syndrome unmasked by rapid conduction. Understanding the full spectrum of tachyarrhythmias and the ACLS approach to managing them is essential for any provider working in high-acuity settings. The Adult Tachycardia with a Pulse Algorithm provides the clinical framework for approaching these rhythms systematically, but remember — the algorithm assumes you have already considered the underlying cause.
Because no single laboratory value confirms thyroid storm — TSH and free T4 levels overlap significantly with uncomplicated hyperthyroidism — the diagnosis is clinical. The Burch-Wartofsky Point Scale (BWPS) assigns points across five domains: thermoregulatory dysfunction, central nervous system effects, gastrointestinal and hepatic manifestations, cardiovascular dysfunction, and the presence of a precipitating event.
A score of 45 or higher is highly suggestive of thyroid storm. A score of 25 to 44 indicates impending thyroid storm. A score below 25 makes thyroid storm unlikely. Critically, treatment should not wait for laboratory confirmation — initiation of therapy based on clinical suspicion is the standard of care. The Burch-Wartofsky score keeps providers anchored to the full clinical picture rather than over-relying on any single finding.
Key point allocations within the BWPS cardiovascular domain reflect the urgency of the situation: tachycardia at 100-109 bpm scores 5 points, 110-119 scores 10, 120-129 scores 15, 130-139 scores 20, and above 140 scores 25 points. Atrial fibrillation adds 10 points independently. Heart failure adds 5 to 15 points depending on severity. According to StatPearls thyroid storm data, a febrile patient with AFib at 150 and mild heart failure can already score in the storm-probable range before CNS and GI findings are even factored in.
Thyroid storm does not typically arise de novo — it is triggered by physiologic stress in a patient with underlying hyperthyroidism, often previously undiagnosed. Common precipitants include infection (most common), surgery, trauma, radioiodine therapy, iodine-containing contrast agents, and medication non-compliance. In the emergency setting, the combination of infection-driven systemic inflammatory response plus underlying undiagnosed Graves disease or toxic multinodular goiter is a dangerous setup. History should include recent illness, iodine contrast exposure, and thyroid medication use. Research published in a cardiovascular events analysis of thyroid storm patients confirms that cardiovascular complications are the leading driver of mortality, reinforcing why rapid recognition and cardiovascular stabilization must occur in parallel.
Management follows a coordinated, multi-drug approach targeting each step of the thyroid hormone synthesis and action pathway. The core pharmacologic approach requires addressing beta-blockade, thionamide therapy, iodine administration, and corticosteroid coverage — in that order, with timing critical to avoid paradoxical worsening.
Beta-blockade is the first pharmacologic priority and directly addresses the cardiovascular storm. Propranolol is the traditional agent of choice at 40-80 mg orally every 4-6 hours or 0.5-1 mg IV slowly. Propranolol is preferred over cardioselective beta-blockers in thyroid storm because it also blocks peripheral conversion of T4 to the more active T3. However, emerging evidence from Japanese guidelines suggests esmolol may be associated with lower mortality in hemodynamically compromised patients due to its titratability and ultra-short half-life. In patients with overt heart failure from high-output state, extreme caution with beta-blockade is warranted — heart failure here is from the tachycardia itself, and rate control may actually improve cardiac function.
Thionamides — propylthiouracil (PTU) or methimazole — block new thyroid hormone synthesis. PTU is favored in the acute setting because it also blocks peripheral T4-to-T3 conversion. The loading dose of PTU is 500-1000 mg followed by 250 mg every 4 hours. Iodine (Lugol solution or SSKI) blocks hormone release via the Wolff-Chaikoff effect, but must be given at least 1 hour after thionamides to prevent the iodine from being used to synthesize more hormone. Hydrocortisone 100 mg IV every 8 hours serves multiple roles: it reduces peripheral T4-to-T3 conversion, covers relative adrenal insufficiency that can accompany thyroid storm, and provides an anti-inflammatory effect. Fever management with acetaminophen — not aspirin, which displaces thyroid hormone from binding proteins — and active cooling complete the supportive regimen. All patients require ICU-level care with continuous cardiac monitoring.
Adrenal crisis — also called acute adrenal insufficiency — occurs when the adrenal glands fail to produce adequate cortisol and, in primary adrenal insufficiency, aldosterone in response to physiologic stress. The resulting hemodynamic collapse is profound and can be clinically indistinguishable from cardiogenic shock, distributive shock from sepsis, or even cardiac arrest from non-cardiac causes. According to StatPearls on adrenal crisis, the incidence of adrenal insufficiency in critically ill patients has been reported at 30-60 percent, making it one of the most underrecognized conditions in the ICU and emergency department.
Cortisol is essential for vascular tone maintenance, catecholamine sensitivity, and myocardial function. Without adequate cortisol, blood vessels lose their responsiveness to epinephrine and norepinephrine — a condition called catecholamine resistance. Vasodilation becomes profound and refractory. Simultaneously, aldosterone deficiency in primary adrenal insufficiency causes sodium wasting, volume depletion, and hyperkalemia. The combined effect is a patient in distributive and hypovolemic shock with a heart that cannot respond normally to resuscitation.
The cardiac manifestations are particularly deceptive. Hyperkalemia from aldosterone deficiency produces ECG changes including peaked T waves, prolonged PR interval, widened QRS, and ultimately ventricular fibrillation or asystole. For providers familiar with the ECG patterns of hyperkalemia-induced cardiac arrest, recognizing that these same changes can occur in adrenal crisis is critical. Our detailed review on Hyperkalemia-Induced Cardiac Arrest: Recognition and Emergency Treatment covers the full spectrum of ECG findings and management strategies that directly apply here.
The clinical presentation of adrenal crisis includes hypotension refractory to IV fluids and vasopressors, tachycardia, nausea, vomiting, abdominal pain, weakness, and altered mental status. The refractory hypotension is what drives providers toward cardiac diagnoses. When a patient does not respond to aggressive fluid resuscitation and requires escalating vasopressor support, the first instinct is cardiogenic shock — left ventricular failure, massive pulmonary embolism, or cardiac tamponade. Adrenal crisis rarely enters the differential unless someone specifically thinks to ask: has this patient ever been on chronic steroids? Do they have a known autoimmune condition? Do they have skin hyperpigmentation consistent with Addison disease?
Cases of adrenal crisis-induced stress cardiomyopathy have been documented in the literature, where the hemodynamic stress of cortisol deficiency triggers a Takotsubo-like cardiomyopathy pattern on echocardiography. According to a published case report on adrenal crisis-induced stress cardiomyopathy, echocardiographic findings can create a compelling argument for primary cardiac disease, further delaying recognition of the true cause. Once hydrocortisone is administered, the cardiomyopathy typically resolves — dramatically illustrating the endocrine etiology.
Adrenal crisis can progress to cardiac arrest. When it does, it falls within the framework of reversible causes that all ACLS providers must systematically evaluate. The Hs and Ts mnemonic — hypovolemia, hypoxia, hydrogen ion acidosis, hypo/hyperkalemia, hypothermia, tension pneumothorax, tamponade, toxins, and thrombosis — provides the scaffold for this evaluation. Endocrine emergencies including adrenal crisis and thyroid storm contribute to multiple H-category derangements simultaneously: hypovolemia, acidosis, and electrolyte abnormalities. Mastery of the Hs and Ts framework is essential for any provider encountering unexplained cardiac arrest or refractory shock. Review the full framework in our article on Sudden Cardiac Arrest: The Hs and Ts You Need to Know.
The laboratory constellation of adrenal crisis provides important diagnostic clues even before cortisol levels return. Classic findings include hyponatremia, hyperkalemia, hypoglycemia, and elevated BUN. Eosinophilia on the CBC can be a subtle hint in the right clinical context. The sodium-potassium ratio below 30 has been described as a simple bedside indicator of primary adrenal insufficiency, though it lacks specificity in isolation.
A random cortisol level below 18 mcg/dL in a critically ill patient is strongly suggestive of adrenal insufficiency, though the cutoff is debated. Crucially, the diagnosis should be a clinical one in the emergent setting — do not wait for cortisol results to treat a patient in refractory shock with compatible features. A random cortisol can be drawn, but treatment with hydrocortisone should not be delayed pending results. If diagnostic uncertainty exists, an ACTH stimulation test can be performed after the crisis is managed, but not during it.
Key clinical history features that should raise suspicion for adrenal crisis include: known Addison disease or primary adrenal insufficiency, chronic corticosteroid use (even inhaled or topical steroids in high doses), recent steroid taper or abrupt discontinuation, history of adrenal surgery or bilateral adrenalectomy, autoimmune conditions such as thyroid disease, type 1 diabetes, and vitiligo clustering, and HIV or tuberculosis which can cause adrenal destruction. The patient who has been on prednisone for rheumatoid arthritis and is now in unexplained shock after a GI illness should always have adrenal crisis on the differential.
The cornerstone of adrenal crisis management is immediate glucocorticoid replacement. Hydrocortisone 100 mg IV should be given as a bolus the moment adrenal crisis is suspected in a hemodynamically unstable patient. This is followed by either continuous infusion at 200 mg per 24 hours or bolus dosing of 50 mg IV every 6 hours. Hydrocortisone at these doses provides both glucocorticoid and sufficient mineralocorticoid activity to address both deficiencies.
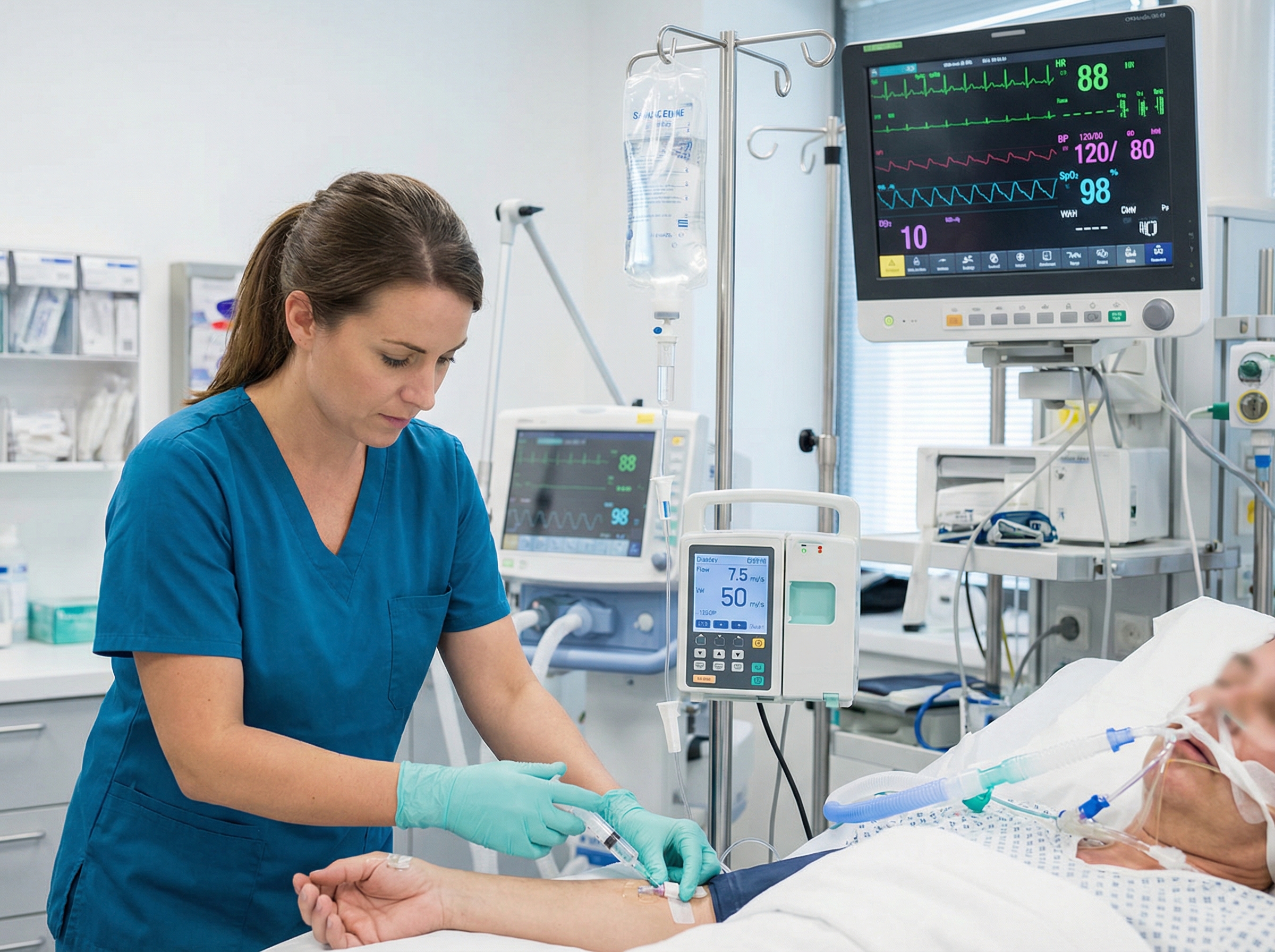
Aggressive volume resuscitation with normal saline is simultaneously required — deficits of 2 to 3 liters or more are common at presentation. Glucose replacement addresses hypoglycemia. Vasopressors may be needed as a bridge, but the critical insight is that these patients often fail to respond to vasopressors until hydrocortisone is on board. Recognizing this pattern — vasopressor-refractory shock that dramatically responds to steroids — is one of the most clinically important observations in emergency medicine. The hemodynamic management principles overlapping here are the same ones covered in advanced life support training, including the management of the bradycardic, hypotensive patient as outlined in the Adult Bradycardia with a Pulse Algorithm.
Mineralocorticoid replacement with fludrocortisone 0.1 mg daily should be initiated once the patient is stabilized and tolerating oral medications, specifically in primary adrenal insufficiency. Secondary adrenal insufficiency — from pituitary or hypothalamic disease — does not require mineralocorticoid replacement since the renin-angiotensin-aldosterone axis remains intact. Identifying and treating the precipitating cause including infection, surgery, or missed medications is equally critical to prevent recurrence.
Both thyroid storm and adrenal crisis share the common feature of presenting with hemodynamic instability and cardiac manifestations that can trigger an ACLS algorithm response without identification of the underlying cause. The following clinical features help distinguish endocrine etiologies from primary cardiac disease:
Advanced Cardiac Life Support training provides more than cardiac arrest algorithms — it teaches the systematic identification of reversible causes that underlie hemodynamic emergencies. Thyroid storm and adrenal crisis both fit within the reversible cause framework as endocrine contributors to the Hs of hypovolemia, acidosis, and electrolyte abnormality. Recognizing these conditions as ACLS-relevant emergencies — not just endocrinology consultations — reframes how providers approach the undifferentiated sick patient.
The ACLS medications that providers use during cardiac arrest — epinephrine, atropine, amiodarone — are the same agents that interact with the dysrhythmias produced by thyroid storm and adrenal crisis. Understanding the pharmacology in context is essential. For instance, epinephrine in a thyroid storm patient already at peak catecholamine sensitivity may exacerbate arrhythmia, while amiodarone carries iodine that could theoretically worsen thyroid excess. These nuances highlight why recognizing the underlying diagnosis early — before cardiac arrest occurs — is far preferable to managing the arrest after the fact. The ACLS Medications Cheat Sheet is an excellent reference for reviewing pharmacologic options and their indications in the context of these complex presentations.
Providers should also recognize PEA arrest as a potential endpoint for both conditions. Both adrenal crisis and thyroid storm can drive pulseless electrical activity through electrolyte derangement, volume depletion, and catecholamine dysregulation. Familiarity with the management of pulseless electrical activity causes and treatment helps providers understand why treating the underlying cause — including administering hydrocortisone during a resuscitation when adrenal crisis is suspected — is as important as any algorithm step.
Emergency providers working in high-acuity environments can sharpen their recognition of these endocrine mimics with the following takeaways:
Recognizing endocrine emergencies that mimic cardiac events requires both deep clinical knowledge and a solid foundation in the systematic approach that ACLS provides. The ability to identify reversible causes, manage complex arrhythmias, and work through undifferentiated hemodynamic instability is exactly what comprehensive ACLS training builds — and what differentiates providers who catch these diagnoses from those who miss them.
At Affordable ACLS, our certification courses are developed by Board Certified Emergency Medicine physicians with over 20 years of frontline experience — the same providers who have managed thyroid storms, adrenal crises, and every cardiac mimic in between. Our ACLS course at just $99 (recertification $89) delivers comprehensive, clinically-grounded training that goes beyond algorithm memorization to build genuine clinical reasoning skills. The course is 100 percent online and self-paced, so busy clinicians — nurses, residents, paramedics, NPs, PAs, and physicians across specialties — can complete certification on their own schedule without missing shifts.
With unlimited exam retakes, immediate digital certification upon completion, and a money-back guarantee, there is no barrier to getting or maintaining the credentials that keep your patients safer. Whether you are an experienced emergency physician looking to recertify or a clinician in any specialty preparing for your next high-acuity encounter, our curriculum provides the clinical depth to handle the cases that surprise you. Call us at 866-655-2157 or email support@affordableacls.com to learn more.
The heart is an end organ. When it behaves erratically — rapid arrhythmias, hemodynamic collapse, high-output failure — it is telling you something is wrong, but not always telling you where the problem originated. Thyroid storm and adrenal crisis are two of the most important systemic conditions that use the cardiovascular system as their primary symptom display, and missing them carries a mortality consequence that catching them early can entirely prevent.
The key to recognition is pattern awareness: fever plus tachyarrhythmia demands thyroid storm be considered; refractory shock plus electrolyte derangement demands adrenal crisis be on the differential. Both diagnoses are treatable with targeted, time-sensitive interventions that work quickly when applied correctly. The same clinical rigor that guides ACLS algorithm execution — systematic, step-by-step, reversible-cause-focused — is exactly what protects patients from these endocrine traps. Train for the rhythms, but always think beyond them.
.jpg)

